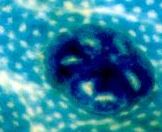
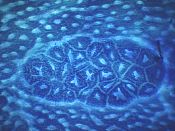
ACF, MDF, BCAC: Pre-Cancerous Lesions?
- ACF are Aberrant Crypt Foci
ACF have been discovered by Ranjana Bird in 1987, in W.R. Bruce's lab in Toronto. Twenty years later, more than 900 articles deal with ACF on PubMed database (Febr. 2007) . Many indirect evidences support the hypothesis that ACF are precancerous lesions ( see ACF as Surrogate Endpoint Biomarkers). The correlation between tumor incidence and number of large ACF (r=0.76, p<0.001, N=36) is a major piece of evidence (full data in Results section, Corpet & Taché, 2002).
However, significant correlation is not the proof of a direct link. In addition, few agents have shown discrepant effects on ACF and on
tumors (namely cholic acid, genistein, raftilose, and 2CPR, a retinoid, table in construction: [ACF/Tumor discrepancies]). Thus, the present study does not prove that ACF are true preneoplastic lesions: may be they never become cancers.
Indeed, the majority of ACF do not turn into cancer, since the azoxymethane dose that yields one cancer per rat yields 100 to 200
ACF per colon. Thirteen years after Bird discovered ACF, other micro-lesions were described: BCAC, MDF, dysplastic ACF, and ACF-min which may
be truly premalignant lesions for colon cancer.
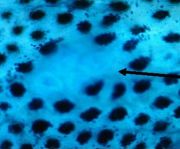
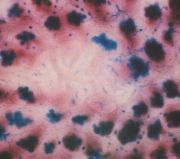
- MDF are Mucin Depleted Foci
MDF were first described by Giovanna Caderni, A. Pietro Femia and Piero
Dolara in 2003. Four years later, 11 articles deal with MDF in PubMed database. Like tumors, but contrary to ACF, MDF are inhibited by synbiotics, an association of
raftilose with lactobacilli (Caderni 2003). Like tumors, but contrary to ACF, MDF are promoted by cholic acid (Femia 2004). MDF may thus be premalignant lesions that predict colon carcinogenesis better
than ACF. Fifteen weeks after injections of azoxymethane to rats (2x15 mg/kg), six to ten MDF
could be scored on the unsectioned colon stained with High-Iron Diamine Alcian Blue (HID-AB). Thus, MDF might be used for chemoprevention studies. Our team have shown MDF promotion by red meat (Pierre et al., J. Nutr. 2004). Most MDF, like tumors, show constitutive activation of the Wnt-signaling pathway with cytoplasmic and nuclear accumulation of beta-catenin
(Femia 2005). Apc mutation was shown by PCR in 6:24 MDF and 7:23 tumors from DMH-injected rats, with similar pattern of nucleotide changes in MDF and tumors: Apc mutations are present in MDF with a frequency similar to that of tumors (Femia 2007). This strengthens the evidence that MDF are precancerous lesions.
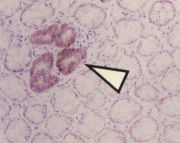
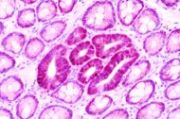
- BCAC are Beta-Catenin-Accumulated Crypts, detected after B-catenin immunostaining of histological thin sections, and first described by Yasuhiro Yamada, Y. Hirose and
Hikedi Mori in 2000
(see Yamada 2000 and Yamada 2001). Five years later, approx. 20 articles deal with BCAC in PubMed database.
A comprehensive discussion of BCAC as a new concept for pre-cancerous lesions for colorectal cancers in
rodents was given by Yamada & Mori (2003).
BCAC growth is enhanced by dietary cholic acid, a known colon tumor promoter, which decreases the number
of ACF in the colon of rats (Hirose 2003). Although BCAC are very appealing as precancerous lesions, they cannot be scored
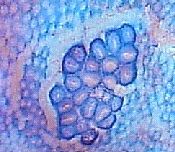
directly on the mucosal surface. They are only identified in the histological sections of en face
preparations (see photos on sides, by Yamada et al., published in Cancer Research).
Thus, BCAC are not yet ready to be used for routine chemoprevention studies, because there
is no practical way to look at the entire colon of a rat histologically.


- DACF, dysplastic ACF resist methanol decolorization after methylene blue staining
Dysplastic ACF (let us call them DACF) were first described in 2005 by Masako Ochiai and Hitoshi Nakagama. After 32 weeks on a PhIP-loaded diet (400 ppm), 8 conventional ACF were observed on the colonic mucosa of F344 male rats. Subsequent decolorization with 70% methanol for 5 min revealed that that 3 of them were DACF: they retained the blue staining, and most of them show dysplasia by histological analysis(Ochiai 2005). Some DACF have non-dysplastic parts, and some of them have are volcano-shaped: tiny-top when seen from above, but wide basis seen on thin section (Nakagama 2005).

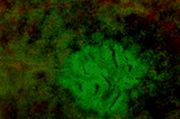
- Flat-ACF or ACF.min are atypical ACF seen in Min mice.
Flat ACF or ACF.min are not elevated above the surrounding mucosa,
but detected by trans-illumination by Paulsen et al. (2000) (photos by J.E.Paulsen, from cited refs). Histopathologic examination of these ACF.min shows displastic crypts. Approximately four ACF.min, but no classical ACF, could be seen in 6-week old mice. When Min mice are
given azoxymethane injections
(2x5mg/kg), both classical ACF and ACF.min can be detected in their colon. But only ACF.min seem to be
associated with the development of adenomas, because (1) they contain dysplastic crypts, (2) they show
overexpression of beta-catenin, and (3) they decrease in number while tumors increase in number ( Paulsen et al., 2001).
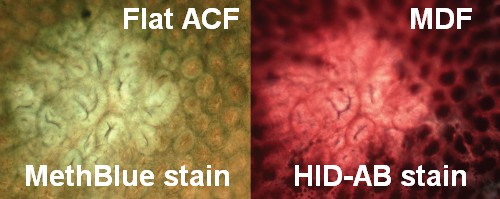 - Flat-ACF are also seen in AOM-injected F344 rats and A/J mice. Flat-ACF overlap with MDF.
- Flat-ACF are also seen in AOM-injected F344 rats and A/J mice. Flat-ACF overlap with MDF.
Flat ACF were found not only in Min mice, but also in AOM-injected rats and mice colon: Flat-ACF have bright blue staining, compressed crypt openings, and crypts not elevated above the surrounding mucosa. Paulsen and Alexander et al. showed that flat dysplastic ACF are related to tumorigenesis in the colon of AOM-treated rats Paulsen et al. (2005) and mice Paulsen et al. (2006): they evidenced continuous developmental growth from small flat dysplastic ACF to the stage of a tumor. These flat-ACF seem more closely related to tumors than classical elevated ACF.
<= In a joint effort, Italian and Norvegian teams scored the same colons to compare Flat ACF and MDF (see photo on the left): They showed that the two lesions overlap was 57% in AOM-injected Min mice colon, and 42% in DMH-injected rats colon Femia et al. (2008).
Corpet's conclusion on ACF, MDF & BCAC
==> In the author's opinion, the most promising early lesions for chemoprevention studies are MDF and flat-ACF.
Up to now, a single laboratory (in Florence, Italy) have published several MDF studies, and a single laboratory (in Oslo, Norway) have published several flat-ACF studies.
In our laboratory, we also were able to score MDF after HID-AB staining (Pierre 2004, Pierre 2008, Santarelli 2010), which confirms Caderni & Femia's findings.
A Japanese team also could score MDF, and showed that they partially overlap with BCAC (Yoshimi 2004). But Yoshimi et al. stained colons with Alcian Blue only (not HID-AB) and their MDF detection is questionable.
Because we had great difficulties in my lab to detect ACF.min in Min mice after AOM injections, we have not yet tried to score flat-ACF. Dyspastic ACF might be useful too, since methanol decolorization is simple to do. However, data are needed to confirm DACF are predictive of cancer outcome, e.g;, sequential studies showing DACF development with time. Last, BCAC clearly seem related to cancer, but there scoring requires histological sections of the whole colon. BCAC chemoprevention studies are thus highly labor intensive and expensive.

It is likely that MDF, BCAC, and Flat-ACF are three views of the same group of lesions (not identical, but largely overlapping Femia et al. (2008)), because
(1) Mucin production lacking in MDF is also usually absent from BCAC (Yamada 2001) and Flat-ACF,
(2) Beta-catenin accumulates in MDF and Flat-ACF cells, as in BCAC and,
(3) The mutation range seems overlapping too (e;g., Ctnnb1 B-catenin gene, and Apc (see paragraphs above).
However, because ACF can easily be scored fast without tissue sectioning, and because they correlate with the tumor outcome, they could remain a useful biomarker for the first screening of agents for chemoprevention [Corpet & Tache, 2002]. We suggest that another surrogate endpoint biomarker be used in addition to ACF, e.g., MDF, flat-ACF or BCAC, to bring a better prediction of tumor and cancer outcome.
MDF ACF BCAC page, a comment to the [Chemoprevention Database].
. Comment homepage.
http://tumor.free.fr -
Author: Denis Corpet - Update 14 june 2010












 - Flat-ACF are also seen in AOM-injected F344 rats and A/J mice. Flat-ACF overlap with MDF.
- Flat-ACF are also seen in AOM-injected F344 rats and A/J mice. Flat-ACF overlap with MDF.